At the Edge of Innovation
Advanced Delivery Systems for U.S. Brands That Refuse to Compromise
R&D and Manufacturing in the USA for oral care, sublingual films, multi-chamber systems, suppositories, topicals, and dietary supplements. learn about our capabilities and find out if we are the right fit for your next project.

Who We Are
We are a US-based contract manufacturer that specializes in difficult, multi-component formulations — from professional in-office teeth-whitening systems engineered for summer heat-stability, to sublingual/orodispersible films (ODF) with high active loads, multi-chamber dispensers, suppository delivery systems, and dietary supplement innovations. What sets us apart is what we are most asked about: our ability to combine rapid prototyping, analytical rigor, and design-for-scale manufacturing to move ideas from lab to launch.
Why U.S Brands Choose Atrium for Complex and Hard-to-Manufacture Projects
Our Core Specialties — Oral Care, Sublingual Films, Suppositories, and Supplements
If you’re expanding your product lines and need a partner who can formulate, scale, and manufacture reliably in the US, you’re in the right place. Atrium Scientific is the best fit for established brands and manufacturers who value Made in USA, cGMP quality systems, and repeatable output.
FDA-Registered Quality Systems and cGMP Compliance at U.S. Manufacturing Scale
Facility: US-based, FDA-registered, cGMP-compliant
QA/QC: Incoming, release testing, stability, retain program, CAPA
Packaging: Oxygen and moisture-barrier materials, dual-chambers, airless, pouches, stick packs, suppositories, capsules, and unit-dose dispensers.
Supply chain: US and vetted global sources, alternate-vendor strategies to protect product launches.
Dietary Supplement Technologies
Atrium specializes in sublingual films, drinkable powders, and capsules, where bad taste or ingredient stability would defeat regular formulations.

Stick Packs
Label-friendly excipients, sugar-free systems, and stability systems.
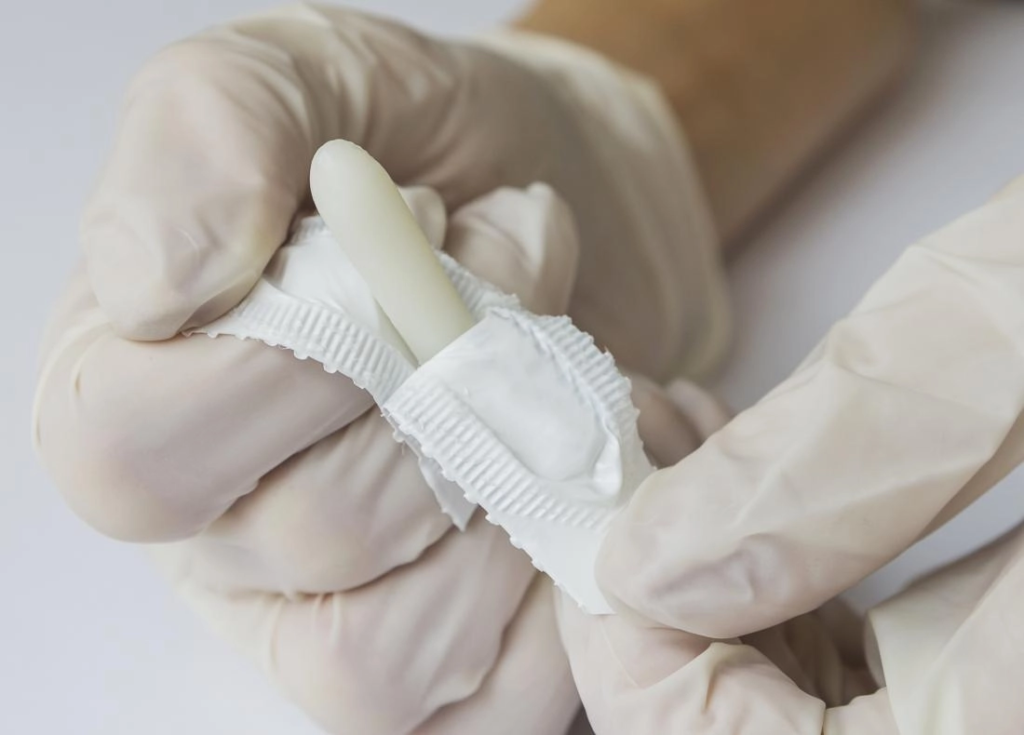
Suppository Delivery Systems
Lipid-based matrices, melting point tuning (33–37 °C), sedimentation control for suspended actives. Release profiling via in-vitro dissolution.
Unit-Dose
Leak-proof unit-dose packaging.

Automation
Fully automated production lines.
Quality
Consistent & strict quality control.


How We Work
From Problem Statement to Launch at US Speed
Problem Framing (Week 0–1): Technical intake, target project summary, claims guardrails, and design for manufacturability (fill sizes, pack lines).
Rapid Prototyping (Weeks 1–6): Formulation sprints; build A/B variants internally around critical vectors – pH, rheology, stability, taste, performance panelist testing.
Analytical & Stress Testing (Weeks 4–8): Assay (titration / HPLC), rheometry (yield stress & thixotropy), accelerated stability (e.g., 50 °C), transport abuse (freeze-thaw), pH drifting, and packaging compatibility analysis (permeation, reaction, stability, etc.).
Scale-Up + IQ/OQ (Weeks 8–10): Pilot batches on coater/gel mixers, line trials on actual pouches/syringes/strips; set CQAs and in-process controls.
Launch (Weeks 10–16): Final specs, CoA, stability plan, artwork/label review; build safety & substantiation documentation.
Case Studies
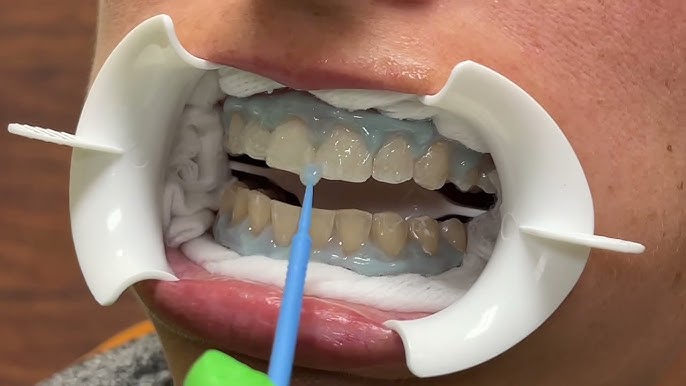
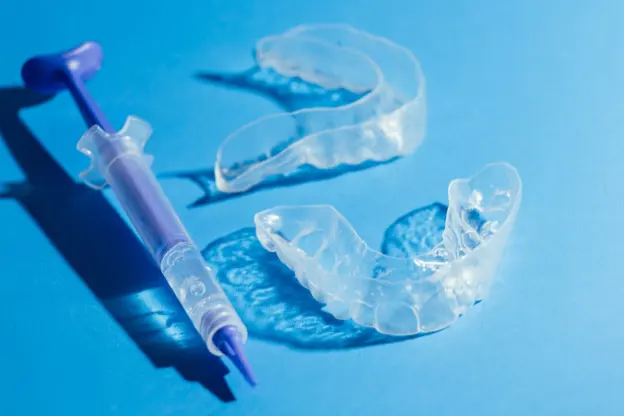
Heat-Stable Professional Whitening Gel (No Cold Shipping Required)
A U.S. in-office whitening brand came to us after hearing about our reputation for solving heat-stability challenges that other manufacturers could not crack. They needed a high-potency peroxide gel that could withstand freight temperatures up to 50°C without potency loss or phase separation. We engineered a water-based system using peroxide-compatible polymers and a radical-scavenger matrix to slow autocatalytic breakdown. The near-neutral pH improved patient comfort and minimized sensitivity. The result: less than 15% peroxide loss after 30 days at 50°C, zero leakage, smooth non-drip chairside handling, and successful U.S. commercialization with repeat orders and a post-launch complaint rate below 0.3%.
High-Load Sublingual Film with Bitter Actives
A brand required a fast-dissolving oral thin film (ODF) delivering >35% active load while effectively masking strong bitter notes. We developed a sublingual film using taste-masking microencapsulation, an optimized PEG plasticizer system, and moisture-protective pouch packaging to preserve stability. The result: disintegration ≤40 seconds (USP <2040>), content uniformity CV ≤5%, and pouch residual moisture ≤3% at 6-month accelerated conditions. The product successfully scaled to continuous roll-to-roll manufacturing with >95% yield post die-cut.
Target Product Summary: claims, channel (professional/DTC/retail), pricing requirements
Functional requirements: actives, flavor, peroxide level (if any), film size / thickness
Performance targets: viscosity, pH, dosage, taste
Packaging preference: syringe/dual-chamber/strip/pouch/suppository
Timelines: prototype date, pilot, first PO
what to
bring
to your
scoping
call
Let’s Build Together
Based in the USA. Fast to sample. Built for scale.
We can start with a feasibility study or a full R&D program.
Email: info@atriumsci.com | Tel: (626) 380-5887
Page last updated: April 15, 2026
Frequently Asked Questions
What does Atrium Scientific Group specialize in?
Atrium Scientific Group is a contract development and manufacturing organization (CDMO) specializing in oral dissolving films (ODF), sublingual strips, suppositories, dietary supplement capsules, teeth whitening strips, and personal care topicals. The company is FDA-registered, cGMP-certified, and operates from a facility in Garden Grove, California (Orange County). Atrium serves supplement brands, oral care companies, DTC businesses, and international brands seeking Made in USA manufacturing.
What makes Atrium Scientific different from other supplement contract manufacturers?
Atrium’s key differentiators are proprietary nano-encapsulation and complexation technology (patented), a low minimum order quantity of 3,000 units across all product formats, and genuine multi-format manufacturing capability under one roof — ODF strips, suppositories, capsules, oral care, and topicals. Leadership includes executives from Johnson & Johnson and polymer science PhD scientists who developed the HarmonyFilm ODF platform. This combination of pharmaceutical science depth, R&D-driven formulation capability, and accessible MOQs is uncommon in the U.S. CDMO market.
Where is Atrium Scientific Group located?
Atrium Scientific Group’s manufacturing facility is located at 10871 Capital Ave, Garden Grove, CA 92843 — in Orange County, California. The facility is FDA-registered, cGMP-compliant, and eligible for client facility audits. Phone: (626) 380-5887 | (714) 858-3097. Email: info@atriumsci.com.
What product formats does Atrium Scientific manufacture?
Atrium manufactures oral dissolving films (ODF) and sublingual strips, rectal and vaginal suppositories, dietary supplement capsules (vegetarian and gelatin), stick packs, teeth whitening strips (PAP and hydrogen peroxide), whitening gels, topical creams and serums, and custom personal care formulations. Both private label (stock formula) and fully custom formulation development services are available.