Custom Rectal and Vaginal Suppository Contract Manufacturing
Suppositories formulated to aid in relief during the period, or for local muscular relaxation and inflammation relief
Plant Extracts in cocoa butter or triglycerides derived from coconut
How Suppositories Benefit the Users
While some users may initially hesitate about suppositories, it’s important to highlight the numerous advantages this dosage form offers. These benefits specifically cater to various groups:
- Elderly: Suppositories are a solution for those who struggle with swallowing.
- People with digestive blockages: Suppositories can bypass obstacles within the digestive tract.
- Individuals experiencing vomiting: When users can’t keep anything down, suppositories provide a reliable alternative.
- Individuals avoiding foul-tasting medicine: For those avoiding the unpleasant taste of certain medications, suppositories offer a welcome alternative.
Unlocking the Power of Harmony™ Suppository Dosage: Revolutionizing Bioavailability
Suppositories are a game-changer in the world of bioavailability:
Enhanced Bioavailability: Suppositories boast increased bioavailability, ensuring efficient absorption by vital tissues.
First-Pass Metabolism Bypass: Unlike ingredients that can be compromised in the gastrointestinal tract, break down too rapidly, or irritate the stomach lining, suppositories take a shortcut, avoiding first-pass metabolism.
Rapid, High-Dose Delivery: Suppositories are the champions of swift and potent medication delivery, eliminating the need for specialized equipment or storage conditions. As the elderly population continues to grow, suppository-based delivery is emerging as a straightforward solution. The market has taken notice of this growing interest. According to an analysis by Allied Market Research, the global suppositories market was valued at $1,410.93 million in 2020 and is projected to reach $2,402.46 million by 2030, reflecting a CAGR of 5.7% from 2021 to 2030. Get ready for a suppository revolution!
Product Features
Experience the soothing, tension-melting effects of our suppositories
Each suppository delivers up to 375 mg of plant extracts (any blends) directly to the area that needs it most
Free of solvents, heavy metals, and pesticides
No additives or preservatives
8X higher bioavailability and absorption rate compared to oral administration (gastrointestinal)
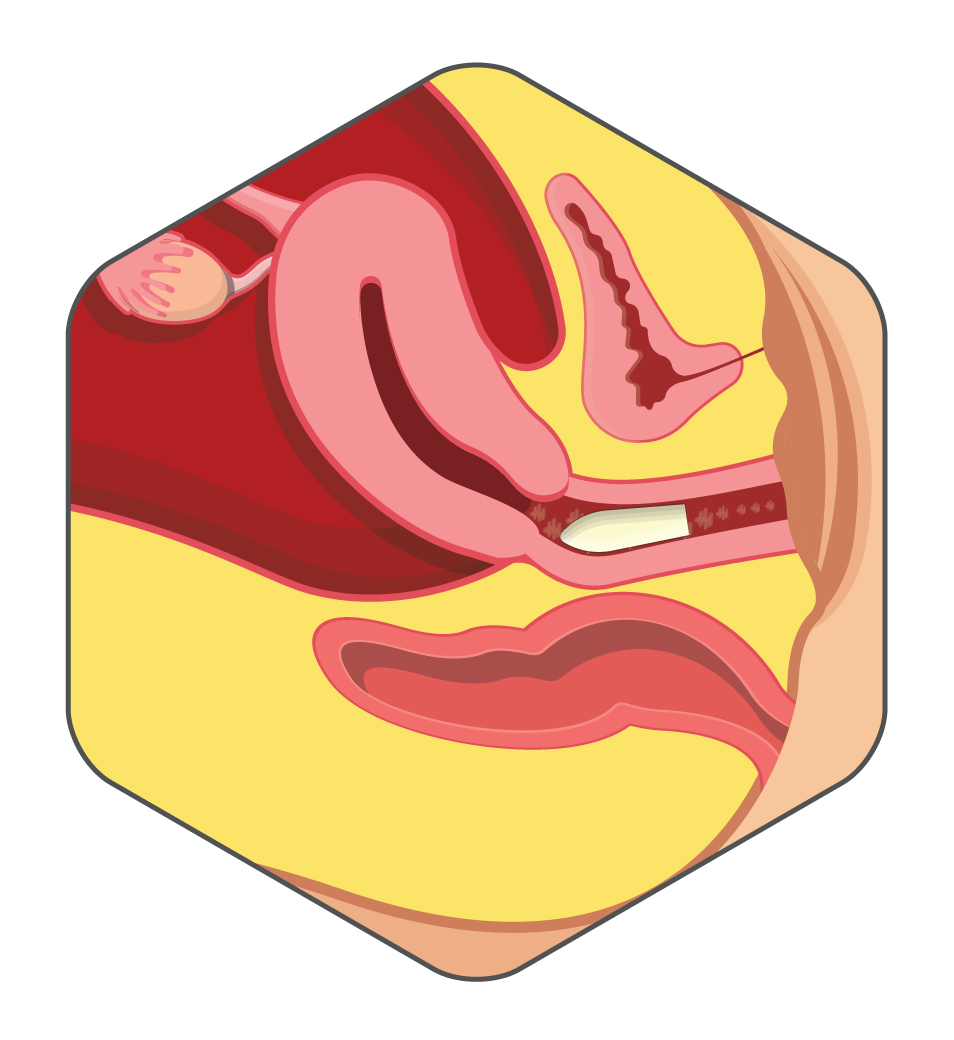
Users find Harmony suppository helpful for improved vaginal comfort and overall wellness
Storage Quirks: Certain suppositories demand special treatment – some need refrigeration in hot climates, while others require unique packaging to avoid moisture mishaps.
Patient Concerns: Practical issues plague patient acceptance, including irritation, leakage, and the overall reluctance to embrace suppositories.
Scientific Puzzles: Developing suppositories requires both scientific precision and formulation expertise. Balancing high dosage, shape, solubility, and stability can be challenging. Additionally, sourcing high-quality materials and controlling manufacturing costs adds another layer of complexity. Research and pharmaceutical guidance from organizations like the World Health Organization often highlight the importance of proper formulation and safe drug delivery systems.
But wait, there’s more to the story:
A Unique Niche: Suppository supplement manufacturing is a niche expertise, and finding the right manufacturer is key. Enter Atrium Scientific, with over 15 million doses produced on cutting-edge equipment.
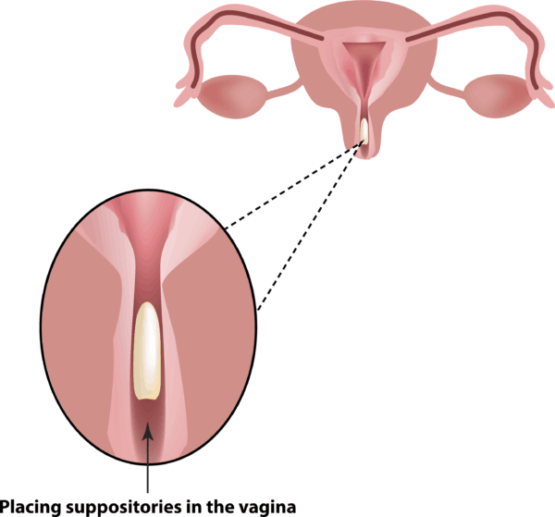
Intriguingly, suppositories offer solutions where other forms fall short, holding untapped potential for unmet needs. Stay tuned for the suppository revolution!
At Atrium Scientific, we focus on delivering science-driven dietary supplement manufacturing solutions that support modern wellness needs. Our advanced production capabilities ensure high-quality formulation, stability, and performance across a wide range of supplement products.
Quality and safety are central to our development process. Following global health guidance from the World Health Organization on nutritional support and safe supplementation, we help brands create reliable products that meet regulatory and consumer expectations. Whether you are planning Harmony™ Suppository, capsules, tablets, or specialized supplement delivery systems, our team can support your project from concept to commercialization.
Explore our advanced manufacturing and formulation capabilities for modern health solutions. Visit our Dietary Supplement Manufacturing page to learn more or discuss your custom project with our team today.
Related services: ODF Sublingual Film Manufacturing • Capsule Manufacturing • Request a Quote
Page last updated: April 18, 2026
Frequently Asked Questions
What types of suppositories does Atrium Scientific manufacture?
Atrium Scientific manufactures rectal and vaginal suppositories for dietary supplement and wellness brands. Product categories include boric acid suppositories, probiotic vaginal suppositories, CBD suppositories, botanical wellness suppositories, and custom formulations for supplement brands targeting rectal or vaginal delivery. Both glycerinated gelatin and hydrogenated vegetable oil (Witepsol) bases are available.
What is the minimum order quantity for suppository contract manufacturing at Atrium?
Atrium Scientific’s minimum order quantity for suppository manufacturing is 3,000 units — one of the lowest MOQs from a U.S.-based, FDA-registered suppository contract manufacturer. This low threshold is designed for supplement brands, wellness companies, and DTC businesses launching their first suppository product without large volume commitments.
What regulatory classification applies to dietary supplement suppositories?
Dietary supplement suppositories — including boric acid, probiotic, and botanical formulations marketed with structure/function claims — are classified as dietary supplements under DSHEA and subject to 21 CFR Part 111 cGMP requirements. Products making disease treatment claims are regulated as OTC drugs. Products marketed for cosmetic wellness purposes may be classified as cosmetics. Atrium’s team provides regulatory guidance on claim positioning and classification during product development.
Can Atrium Scientific develop a custom suppository formula?
Yes. Atrium offers full custom suppository formulation development from concept through commercial production. Services include base selection, active ingredient incorporation, prototype manufacturing, stability testing, packaging validation, and process scale-up. Custom formulas developed for a brand are proprietary to that brand under IP provisions in the manufacturing agreement. Typical timelines for custom suppository development run 16 to 24 weeks from concept to first production run.
Does Atrium’s suppository manufacturing facility support international export?
Yes. Atrium’s FDA-registered, cGMP-certified facility in Garden Grove, California provides manufacturing documentation supporting export to Australia (TGA GMP recognition), Canada (Health Canada NHP documentation), and other markets requiring FDA-registered facility certification. International brands receive the same manufacturing standards and documentation support as domestic clients.


