The Science Behind Transmucosal Drug and Supplement Delivery
Transmucosal delivery — the absorption of active compounds through mucosal membranes rather than the gastrointestinal tract — is one of the most clinically well-characterized alternative routes for both pharmaceutical and nutraceutical actives. For supplement brands evaluating oral dissolving films, understanding the underlying science is not merely academic. It provides the mechanistic foundation for bioavailability claims, helps identify which actives are genuine candidates for transmucosal delivery, and guides formulation decisions that determine whether a product delivers on its promise. This article covers the anatomy, pharmacokinetics, formulation science, and practical limitations of transmucosal supplement delivery at a level of technical depth useful to product developers and informed brand owners.
Oral Mucosal Anatomy: The Foundation of Transmucosal Delivery
The oral mucosa is a continuous epithelial lining covering the interior of the mouth. It is not structurally uniform — four anatomically and functionally distinct regions exist within the oral cavity, each with different epithelial thickness, surface area, keratinization status, and vascular supply. These differences translate directly into dramatically different absorption characteristics.
Sublingual Region
The floor of the mouth beneath the tongue is the most pharmacokinetically favorable mucosal region for rapid systemic delivery. The sublingual epithelium is non-keratinized, thin (100–200 micrometers), and richly vascularized with a dense capillary plexus in the underlying lamina propria. Saliva production in this region maintains a moist environment that facilitates rapid film dissolution. The combination of thinness, non-keratinization, and high vascularity makes the sublingual mucosa the fastest and most efficient absorption site in the oral cavity for appropriately formulated actives.
Buccal Region
The inner surface of the cheeks presents a significantly thicker non-keratinized epithelium (500–800 micrometers) with a lower vascular density than the sublingual region. Absorption through the buccal mucosa is slower and more sustained than sublingual delivery, making it better suited to applications requiring prolonged mucosal contact and controlled release. The larger surface area of the buccal region (approximately 50 cm² per cheek) partially compensates for its lower permeability compared to the sublingual floor.
Gingival and Palatal Regions
The gingival mucosa (gums) and hard palate are partially to heavily keratinized — a protective adaptation to mechanical stress from chewing and food contact. Keratinization significantly reduces permeability, making these regions poor absorption sites for systemic delivery. They are not relevant targets for ODF supplement formulations.
The Vascular Drainage Pathway: Why Transmucosal Delivery Bypasses First-Pass Metabolism
The pharmacokinetic advantage of transmucosal delivery over swallowed oral forms is rooted in the venous drainage anatomy of the oral cavity. Compounds absorbed through the sublingual mucosa enter the capillaries of the lamina propria and drain into the sublingual and deep lingual veins, which merge into the lingual vein and subsequently the internal jugular vein. From the internal jugular, blood flows to the superior vena cava and directly into systemic circulation.
This pathway entirely bypasses the portal venous system — the dedicated circulatory route that carries blood from the gastrointestinal tract to the liver before systemic distribution. The liver’s first-pass metabolic processing of swallowed compounds, which can reduce bioavailability of high-extraction actives by 50–85%, is therefore circumvented by the transmucosal route. For actives with high hepatic extraction ratios, this bypass represents a substantial pharmacokinetic advantage that cannot be replicated by dose escalation in swallowed oral forms.
Physicochemical Factors Governing Mucosal Permeability
Not all active compounds cross the oral mucosa with equal efficiency. Whether transmucosal delivery provides a meaningful bioavailability advantage for a specific active depends on the interplay of several physicochemical properties that determine how readily the molecule crosses the mucosal epithelial barrier.
Molecular Weight and Size
Passive diffusion across the oral mucosa — the dominant absorption mechanism for most supplement actives — is strongly size-dependent. Small molecules below approximately 600 Daltons cross mucosal membranes efficiently by both transcellular (through cell membranes) and paracellular (through intercellular junctions) pathways. Above this threshold, passive diffusion rates fall sharply. This molecular weight ceiling is a primary limiting factor for the transmucosal delivery of peptides, proteins, and larger polysaccharides, which require permeation enhancement strategies or alternative delivery approaches.
Lipophilicity (Log P)
The oral mucosal epithelium, like all biological membranes, is a lipid bilayer structure. Lipophilic compounds (positive log P values) partition readily into cell membrane lipids and cross by transcellular diffusion. Hydrophilic compounds rely more heavily on paracellular transport through intercellular tight junctions, which are more restrictive. Compounds with log P values between approximately 1 and 3 typically show the best balance of aqueous solubility (for dissolution in saliva) and membrane permeability. Highly hydrophilic actives (log P below 0) show limited passive transmucosal absorption without enhancement strategies.
Ionization State (pKa and Salivary pH)
Biological membranes are selectively permeable to non-ionized molecular species. The ratio of ionized to non-ionized forms of an active compound at any given pH is governed by the Henderson-Hasselbalch equation and the compound’s pKa. Salivary pH ranges from approximately 6.2 to 7.4 depending on flow rate and individual variation. For weak acids and weak bases with pKa values in or near this range, a significant fraction of molecules will be in the non-ionized, membrane-permeable form at salivary pH — a prerequisite for efficient passive transmucosal absorption.
Protein Binding and Tissue Binding
Compounds with high plasma protein binding have a reduced free fraction available for distribution after absorption, though this is a systemic pharmacokinetic consideration rather than a determinant of mucosal absorption efficiency per se. Tissue binding within the mucosal epithelium itself — relevant for certain cationic compounds — can create a depot effect that slows permeation and reduces peak absorption rate.
How ODF Films Facilitate Transmucosal Delivery
Oral dissolving films are not simply a convenient delivery form — their pharmaceutical engineering directly enables and optimizes the transmucosal absorption process in ways that alternatives such as sublingual tablets or tinctures cannot match.
Polymer Matrix and Dissolution Kinetics
The film-forming polymer determines how quickly and completely the strip dissolves in saliva, releasing the active ingredient in dissolved form for mucosal contact. Rapidly dissolving polymers such as pullulan release the active within 15–30 seconds, maximizing the concentration gradient at the mucosal surface during the critical absorption window before saliva wash removes the dissolved active. Slower-dissolving HPMC formulations can maintain a dissolved active concentration at the mucosa for several minutes, which may improve absorption of moderately permeable actives by extending contact time.
Mucoadhesion
Certain polymers used in ODF formulations exhibit mucoadhesive properties — they physically adhere to the mucosal surface through hydrogen bonding and electrostatic interactions with mucin glycoproteins. Carbopol, sodium carboxymethylcellulose, and certain grades of HPMC all show mucoadhesive behavior. Incorporating mucoadhesive polymers into a sublingual or buccal film prolongs contact time between the dissolved active and the absorptive surface, improving flux across the mucosa for actives with moderate permeability. This is particularly valuable in buccal film applications where extended residence time is the design objective.
Permeation Enhancers in ODF Formulations
For actives with intrinsically limited transmucosal permeability, food-grade permeation enhancers can be incorporated into the film matrix to transiently and reversibly increase membrane permeability. All permeation enhancers used in dietary supplement ODF formulations must have GRAS status or established food additive regulatory acceptance. Common approaches include:
- Medium-chain fatty acids (caprylic acid, capric acid, lauric acid): Disrupt tight junction integrity and increase paracellular permeability transiently; well-characterized GRAS status; widely used in food applications
- Cyclodextrins (beta-cyclodextrin, hydroxypropyl-beta-cyclodextrin): Form inclusion complexes with lipophilic actives, improving aqueous solubility and mucosal contact without membrane disruption; also useful for taste masking
- Non-ionic surfactants (polysorbate 80, Cremophor EL): Interact with membrane lipids to temporarily increase fluidity and transcellular transport; used at concentrations well below irritation thresholds
Specific Actives: Transmucosal Delivery Evidence
The clinical and pharmacokinetic literature provides well-characterized data for several supplement actives of direct relevance to nutraceutical ODF development.
Melatonin
Melatonin is the most extensively studied supplement active in the context of sublingual delivery. Its oral bioavailability from swallowed tablets is approximately 15% due to extensive first-pass hepatic metabolism. Sublingual melatonin achieves significantly higher and more consistent peak plasma concentrations, with pharmacokinetic studies demonstrating onset within 5–15 minutes — well within the sleep-induction window. The dose-response relationship for sublingual melatonin also suggests effective sleep support at doses as low as 0.1–0.3mg, compared to the 0.5–5mg doses common in swallowed formulations.
Vitamin B12 (Methylcobalamin)
Gastrointestinal absorption of B12 is uniquely limited by intrinsic factor — a glycoprotein secreted by gastric parietal cells that is required for active transport of B12 in the ileum. This mechanism saturates at approximately 1–2 mcg per dose, regardless of oral dose size. Sublingual B12 bypasses intrinsic factor dependency entirely, achieving absorption through passive transmucosal diffusion. Clinical studies have demonstrated that sublingual methylcobalamin at doses of 500–1000 mcg can normalize serum B12 levels in deficient patients, including those with pernicious anemia and gastric absorption impairment, supporting its use as an alternative to intramuscular injection.
Cannabidiol (CBD)
CBD is a lipophilic compound (log P approximately 6.3) with significant first-pass hepatic metabolism when swallowed. Published pharmacokinetic studies report oral bioavailability ranging from 6% to 19%, with high food-dependent variability. Sublingual administration improves both absolute bioavailability and inter-individual consistency by bypassing hepatic first-pass processing, though CBD’s high lipophilicity also means it partitions into mucosal tissue lipids, creating a local depot that can slow peak absorption compared to more hydrophilic actives.
Testosterone and Steroid Hormones
While not a dietary supplement application, the pharmaceutical use of sublingual testosterone demonstrates the pharmacokinetic principle most clearly. Oral testosterone undergoes near-complete first-pass metabolism when swallowed, making standard oral administration therapeutically ineffective. Sublingual testosterone achieves systemic levels adequate for clinical use — a demonstration of the magnitude of first-pass bypass achievable through the sublingual route that provides a conceptual anchor for understanding the bioavailability advantages across less extremely metabolized supplement actives.
Limitations of Transmucosal Delivery
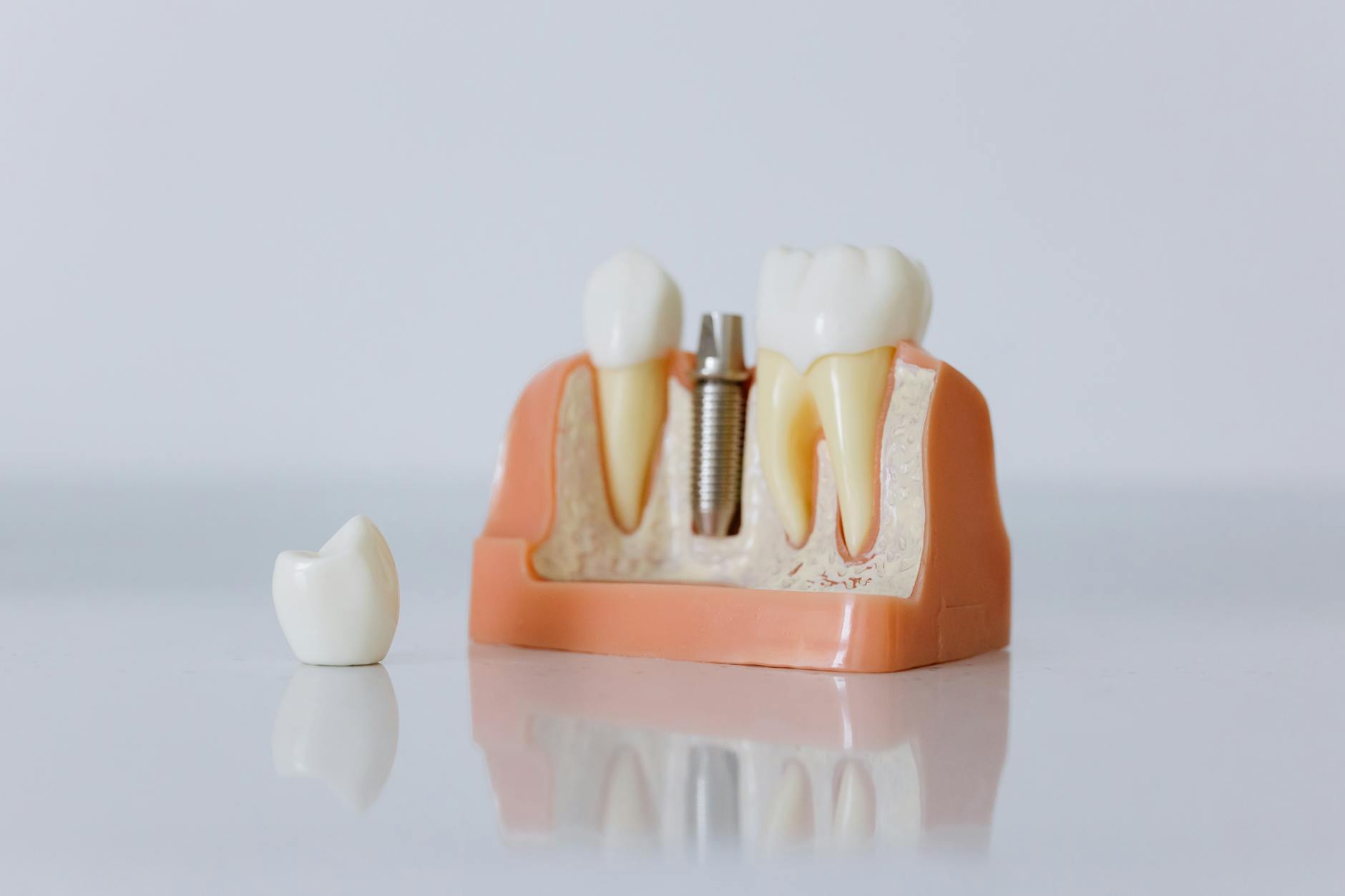
Scientific accuracy requires acknowledging the genuine constraints of transmucosal delivery alongside its advantages. These limitations are not reasons to avoid the format — they are parameters that define appropriate use cases.
| Limitation | Practical Implication | Mitigation Strategy |
|---|---|---|
| Molecular weight ceiling (~600 Da) | Large peptides, proteins, and polysaccharides have minimal transmucosal absorption | Select low-MW bioactive fractions; use permeation enhancers for borderline MW actives |
| Dose ceiling per strip | Typically 1–50mg per strip; high-dose actives not feasible | Multi-strip dosing; reserve ODF for potent, low-dose actives |
| Bitter and unpleasant actives | Direct tongue contact exposes taste receptors; poor palatability reduces compliance | Cyclodextrin complexation, ion exchange resins, flavor and sweetener systems |
| Moisture sensitivity | Hygroscopic films degrade without adequate moisture barrier packaging | Individual foil sachets; validated packaging stability testing |
| Saliva wash | Saliva flow removes dissolved active from the mucosal surface, limiting contact time | Mucoadhesive polymers; rapid-onset absorption before clearance |
How Atrium Scientific Group Applies Transmucosal Science
Atrium Scientific Group’s formulation team applies the physicochemical principles of transmucosal delivery to each ODF development project — evaluating active ingredient permeability, selecting polymer systems appropriate for the target onset profile, developing taste masking strategies for challenging actives, and validating absorption and stability performance from pilot through commercial scale. Our USA-based cGMP facility supports the full ODF development cycle for dietary supplement and nutraceutical brands. Contact our formulation team to discuss how transmucosal delivery science applies to your specific active ingredient and product concept.
Frequently Asked Questions
What is the molecular weight cutoff for effective transmucosal absorption?
Passive transmucosal diffusion is most efficient for molecules below approximately 600 Daltons. Above this threshold, absorption efficiency decreases sharply. Most common supplement actives — amino acids, vitamins, small botanical alkaloids and flavonoids, zinc and other minerals — fall well within this range. Permeation enhancers can extend the practical upper limit somewhat for borderline molecules.
Is transmucosal absorption affected by what the consumer has eaten or drunk recently?
Unlike gastrointestinal absorption, which is significantly affected by food intake (gastric emptying rate, bile acid secretion, pH changes), transmucosal absorption is relatively food-independent. This is an additional consistency advantage over swallowed forms for actives where food-state variability is a concern — CBD being a prominent example where food intake dramatically affects oral bioavailability but has lesser impact on sublingual delivery.
Can the same active ingredient be formulated for both sublingual and buccal delivery?
Yes, but the formulation optimization differs. Sublingual delivery favors rapidly dissolving, low-mucoadhesion films that release the active quickly for fast absorption. Buccal delivery benefits from mucoadhesive polymers that prolong contact time and slower dissolution profiles for sustained release. The appropriate target depends on whether rapid onset or prolonged delivery is the therapeutic or functional objective for your specific product.
How does transmucosal delivery compare to transdermal delivery?
Both routes bypass first-pass hepatic metabolism, but they differ substantially in onset kinetics and suitable actives. Transdermal delivery through skin is significantly slower (hours to reach peak plasma levels) due to the stratum corneum barrier. Transmucosal sublingual delivery achieves peak absorption in minutes. Transdermal is better suited to sustained systemic delivery over hours to days; sublingual is better suited to rapid-onset applications. The two routes are complementary rather than competing for different product design objectives.
Are there any safety concerns specific to transmucosal ODF supplements?
Transmucosal delivery of dietary supplement actives at typical label doses presents no safety concerns beyond those associated with the actives themselves. The mucosal effects of permeation enhancers at food-grade concentrations are transient and reversible. Consumers with oral mucosal conditions (ulcers, inflammation, oral mucositis from chemotherapy) may experience altered absorption or irritation and should consult a healthcare provider. Standard dietary supplement labeling requirements apply regardless of delivery route.
Conclusion
Transmucosal delivery is a pharmacokinetically well-grounded approach to supplement delivery that offers genuine, measurable advantages for appropriately selected actives — advantages rooted in anatomy, biochemistry, and decades of pharmaceutical research. For supplement brands building products where onset speed, bioavailability consistency, and consumer compliance matter, this science provides a credible foundation for differentiated product positioning. The formulation engineering required to realize these advantages in a commercial ODF product is specialized but achievable with the right manufacturing partner. Atrium Scientific Group’s team brings both the scientific expertise and the manufacturing infrastructure to support that development process. Contact us to discuss your next formulation project.
Developing a transmucosal supplement product? Atrium Scientific Group manufactures oral dissolving film strips for sublingual and buccal delivery using GRAS-status excipients and in-house analytical testing. Speak with our formulation team about your transmucosal delivery project.
Page last updated: April 18, 2026
For more context, see our guide on oral dissolving film contract manufacturing.